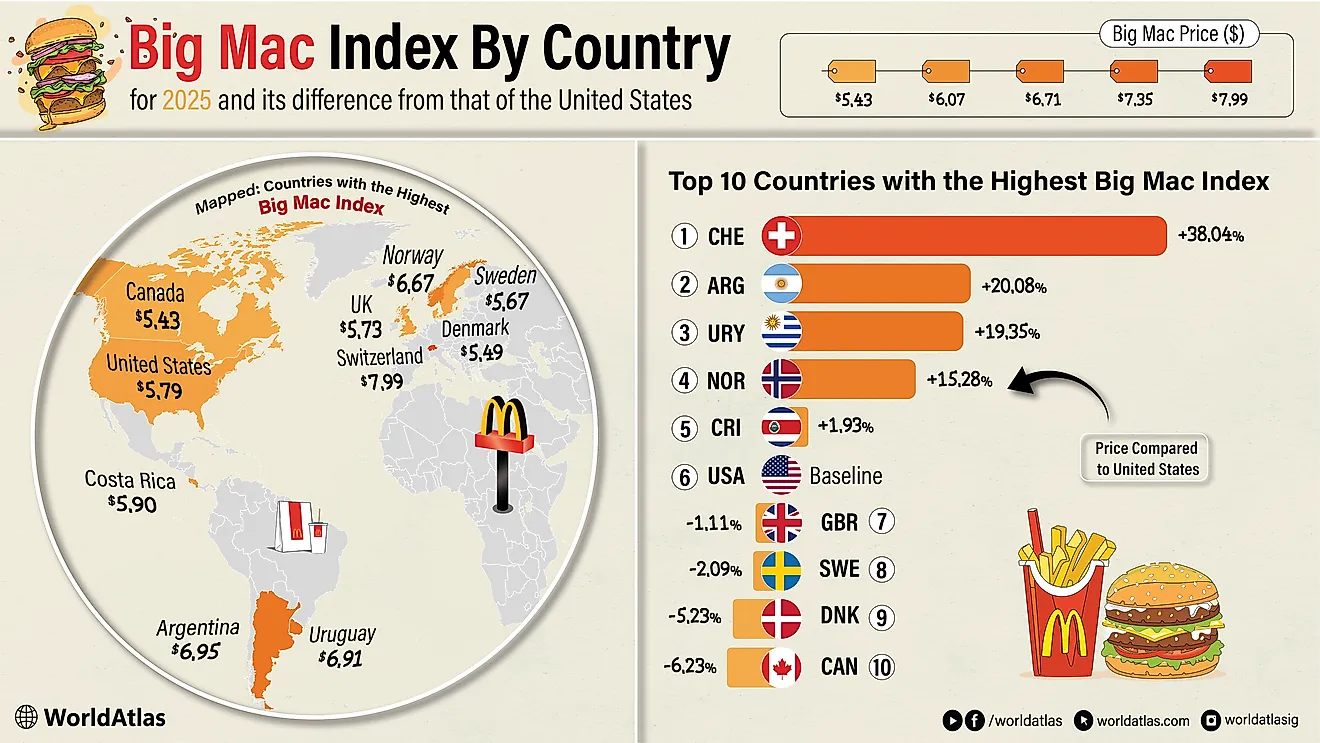
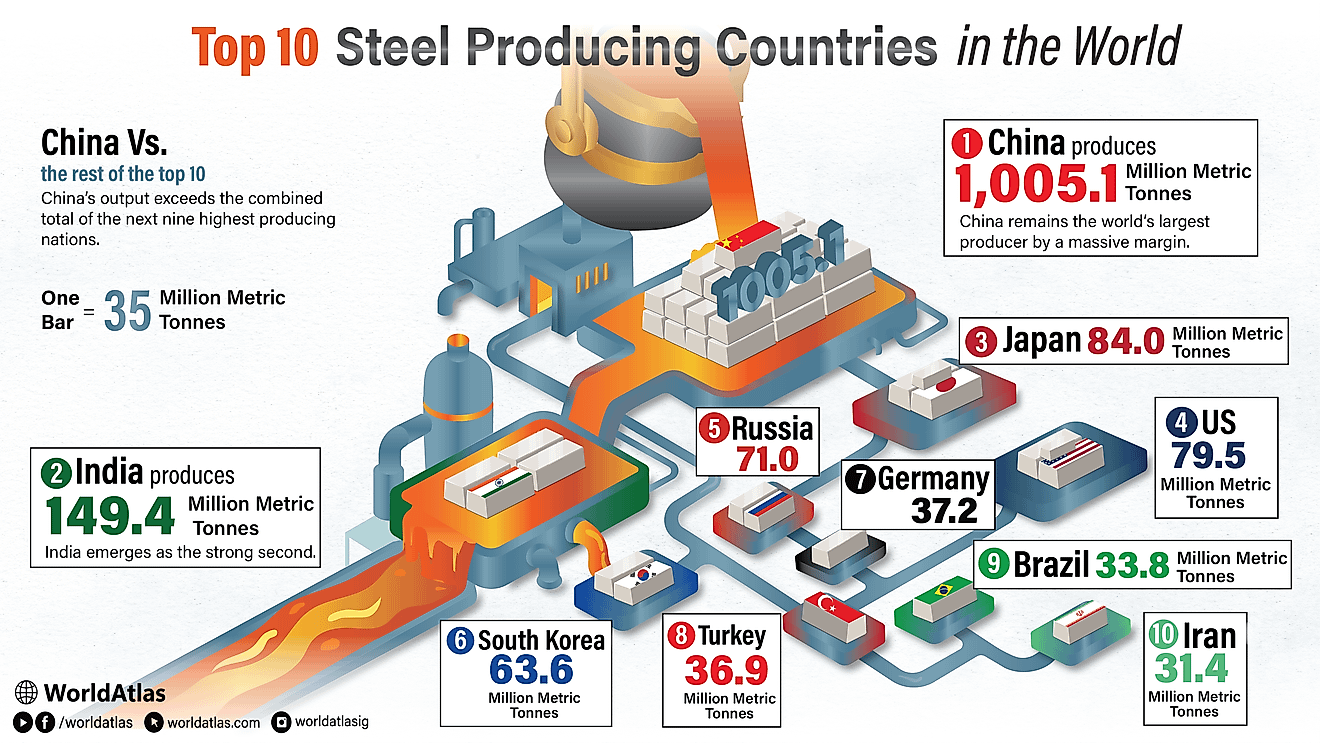
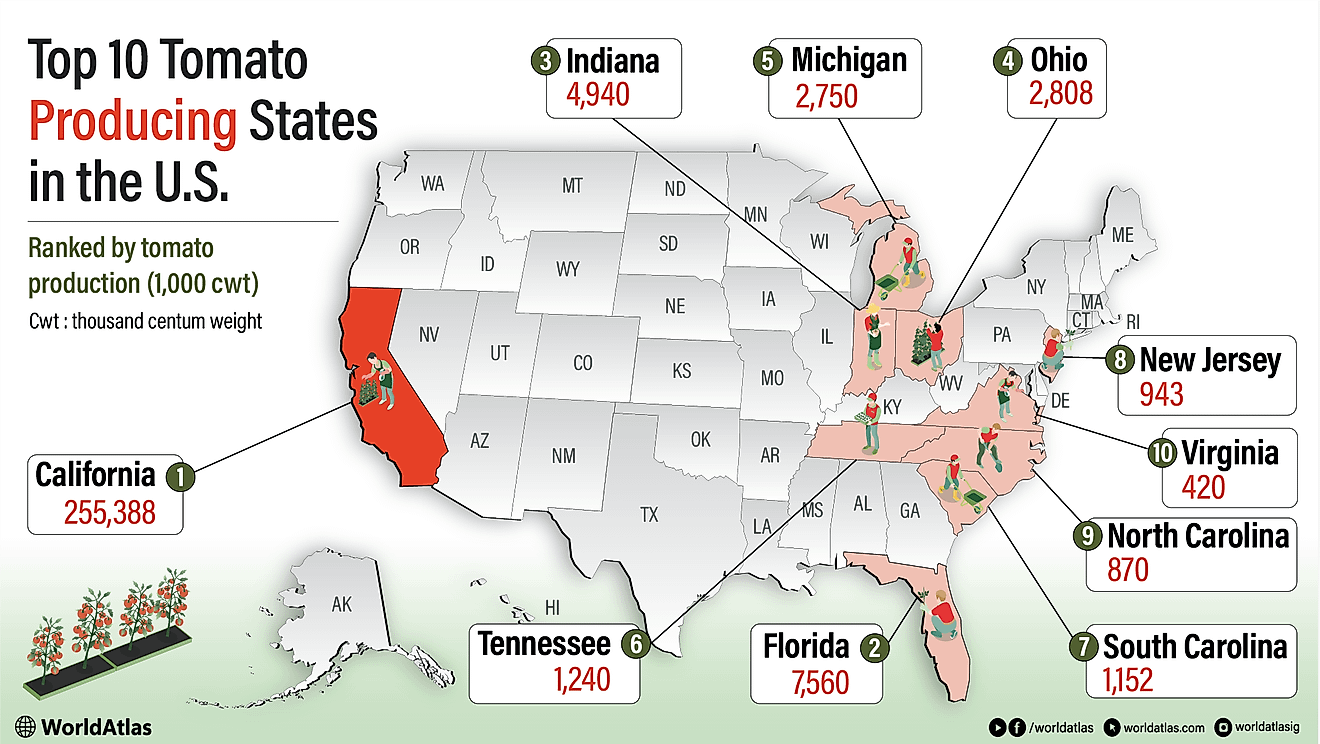
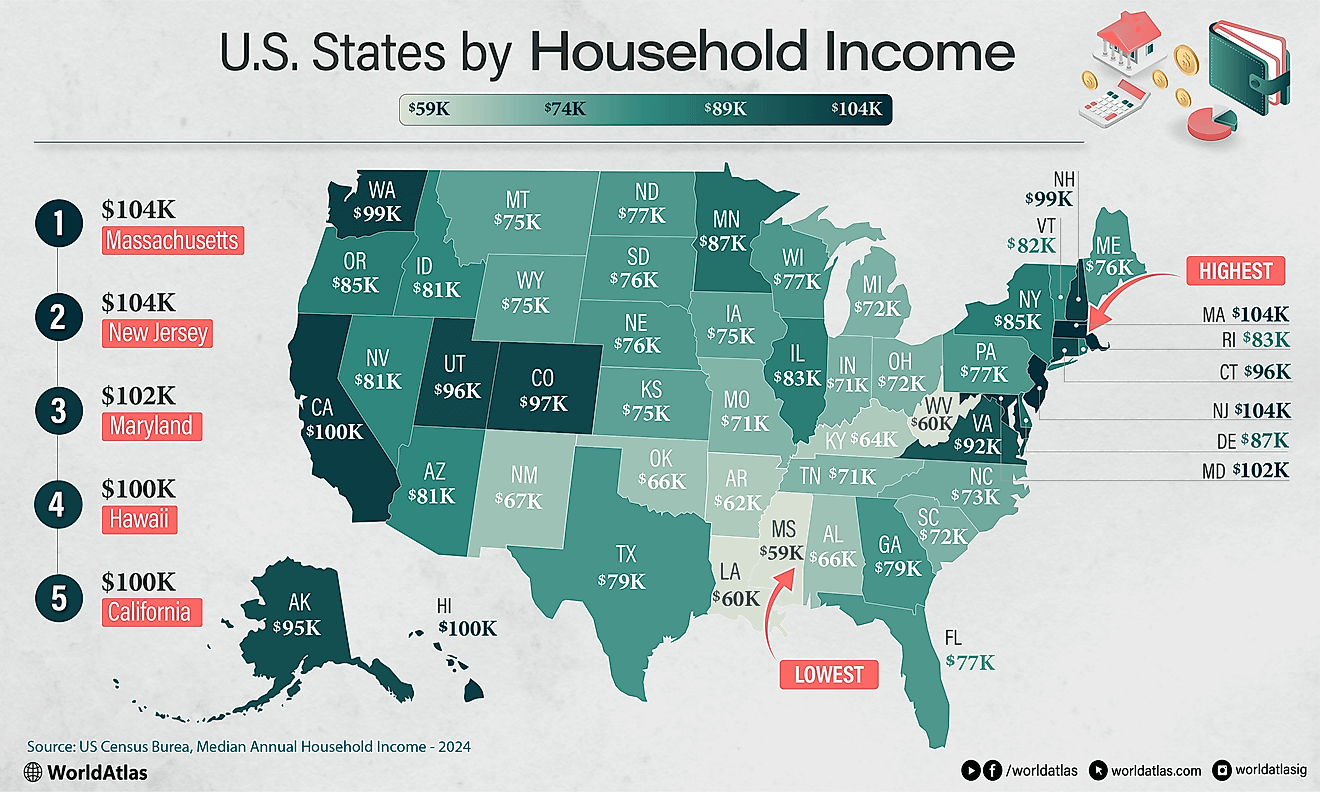
Leading Exporters Of Barium Salts, Barytes, And Witherite

Baryte, also known as barite, is a mineral typically found in cluster as thin to thick sized tabular crystals. Baryte is usually white or colorless, but can also be other colors as well, including yellow, brown, blue, or grey. Baryte is the main source of barium. The most commonly naturally occurring minerals of barium are barium sulphate (barytes) and barium carbonate (witherite). Barytes are somewhat common minerals but witherite is an extremely rare mineral on earth. Now that the basics of these two barium salts have been discussed, this article will analysis the process of extracting, processing and exporting them.
The Process of Exporting Barium
Extraction
Mining methods used to extract baryte vary depending on the type, shape and depth of the deposit. Major bed deposits are usually mined using the open pit method. The baryte is drilled and blasted with explosives and then taken out using front end loaders and other heavy industry vehicles. Vein deposits of baryte are mined using shafts, horizontal passages and sometimes shallow open pits. Residual deposits of baryte are worked on by hand due to the irregular nature of baryte mineralization. Witherite is usually found in fairly pure form and is associating with baryte or other minerals in low temperature hydrothermal environments. Since this is the case witherite is extracted along with these other minerals. Regardless of the extraction method used the mined baryte and witherite are then loaded onto trucks to be taken to factories or labs for processing.
Processing
Since baryte is highly impure and has contaminants, such as waste rock and other minerals, it is processed after being mined. The baryte is separated from these contaminants by gravity separation, wet grinding and froth flotation. Then the baryte is dried. Baryte is turned into barium sulphate by the process of carbothermal reaction, which means that it is heated with coke. For highly pure barium sulphate to be produced barium sulfide is treated with sulfuric acid or salts. Barium carbonate is made commercially and in labs by treating barium sulfide with sodium carbonate using the soda ash method. The barium carbonate that is created from this process is then filtered, washed and dried to finish processing it.
Shipping
Barium sulphate is a health hazard to the eyes and respiratory system, can give off toxic fumes in a fire and can react with aluminum powder to create an explosion. When being handled it protective gloves, googles and clothing should be worn to prevent contact. It should be stored in tightly closed containers, in a well ventilated area away from materials it could react with. It is usually transported via truck, rail or in tanks and should be stored at 77°Fahrenheit (25° Celsius). Barium carbonate is a health hazard is swallowed, can give off toxic fumes in a fire and can react with acids to generate a fire. Protective gear should also be used when handling it. It should be separated from any food, foodstuffs, acids and bromotrifluoride when being transported. It is transported via truck, rail, boat or airplane and is stored in a similar way to barium sulphate.
Applications
Barium sulphate is mostly used as a component in drilling fluid for oil wells. It is used for this since it adds to the density of the drilling fluid, which reduces the chance of a blowout of oil happening from the well. Barium sulphate is also one of the components that makes up white pigment for paints, as a coating for certain photogenic papers and as a filler for plastics. It is also used as a radiocontrast agent when in suspension for x-ray imaging and in green pyrotechnic formulas. Barium carbonate is mostly used in the glazes for the ceramics industry since is acts as a flux and can produce unique colors. It is also added to clay to precipitate away salts that would coat the clay's surface.
Importers of Barium and Its Variants
According to the Observatory of Economic Complexity (OEC) the world's top importer of barium sulphate (barytes) is the United States, who imports 26% of the world's barium sulphate. Rounding out the world's top five barium sulphate importers are Saudi Arabia (9.5%), Canada (4.6%), Netherlands (3.6%) and Germany (3.2%). The top importers in regions of the world not mentioned are Argentina (2.9%) for South America, Algeria (1.3%) for Africa and Australia (0.71%) for Oceania. The world's top importer of barium carbonate (witherite) is Italy, who imports 17% of the world's barium carbonate. Rounding out the world's top five barium carbonate importers are France (12%), Brazil (8.5%), South Africa (6.6%) and India (5.2%). The top importers in regions of the world not mentioned are Mexico (4.8%) for North America and Australia (0.83%) for Oceania.
Leading Exporters Of Barium Salts, Barytes, And Witherite
| Rank | Country | 2015 Barium Salt Exports (USD) |
|---|---|---|
| 1 | China | $272,481,000 |
| 2 | Morocco | $122,359,000 |
| 3 | India | $72,071,000 |
| 4 | United States | $56,910,000 |
| 5 | Netherlands | $33,431,000 |
| 6 | Turkey | $31,471,000 |
| 7 | Kazakhstan | $30,850,000 |
| 8 | Mexico | $27,821,000 |
| 9 | Thailand | $19,014,000 |
| 10 | Spain | $16,737,000 |