The Most Abundant Elements In The Earth's Crust
The Earth's Crust
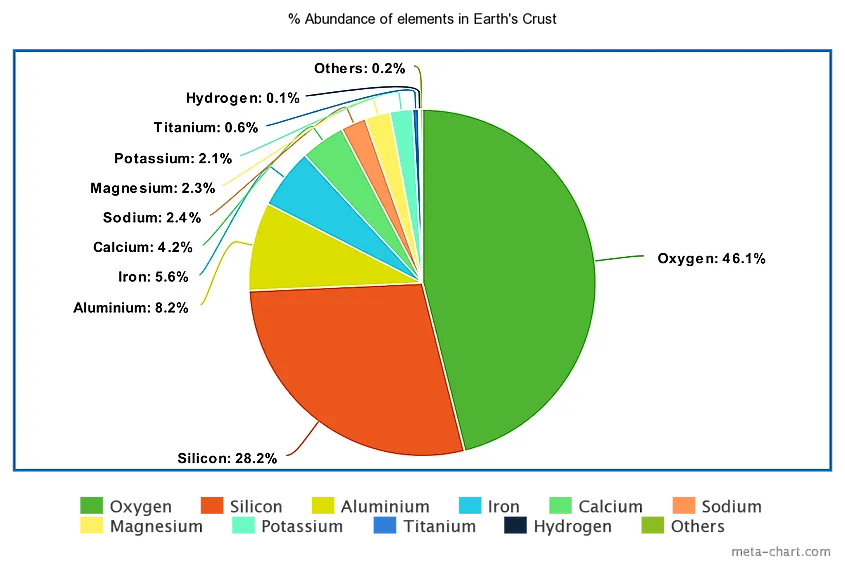
Just two elements (oxygen and silicon) make up roughly 74.3% of the Earth's crust by weight, and the top ten together account for around 99%. Everything else, all 68 of the trace elements ranked below the top ten, combines for less than 1% of crustal mass. The crust itself is the outermost layer of the planet, varying from about 5 to 10 km under the oceans to 30 to 50 km under the continents (commonly averaged at around 40 km), and it makes up only about 1% of Earth's total volume. The percentages below come from the CRC Handbook of Chemistry and Physics, 97th edition. For the prevalence of all 78 elements, scroll down to the full table.
10 Most Abundant Elements In Earth's Crust
| Rank | Element | Abundance (percent by weight) |
|---|---|---|
| 1 | Oxygen | 46.1% |
| 2 | Silicon | 28.2% |
| 3 | Aluminum | 8.23% |
| 4 | Iron | 5.63% |
| 5 | Calcium | 4.15% |
| 6 | Sodium | 2.36% |
| 7 | Magnesium | 2.33% |
| 8 | Potassium | 2.09% |
| 9 | Titanium | 0.565% |
| 10 | Hydrogen | 0.140% |
1. Oxygen (O)
Oxygen is the most abundant element in the Earth's crust at 461,000 parts per million, or about 46.1% by weight. Across the universe as a whole, oxygen ranks third in abundance behind hydrogen and helium. It also makes up roughly 21% of Earth's atmosphere, about 89% of the mass of water, and around 65% of the human body by mass, which makes it the most plentiful element in living tissue. The reason oxygen turns up in so many rocks is that it bonds easily with almost everything else: silicate minerals (combinations of silicon and oxygen) form most of the continental crust, and iron oxides such as hematite and magnetite account for much of the iron the crust contains. Liquid oxygen serves as the oxidizer in cryogenic rocket propellants, combining with fuels like liquid hydrogen or kerosene during launch. Oxyacetylene torches, which mix oxygen with acetylene gas, produce flames around 3,500°C, hot enough for welding and metal cutting.
2. Silicon (Si)
Silicon makes up about 28.2% of the crust by weight, and it almost always appears bonded to oxygen rather than on its own. Silicon dioxide (SiO₂) is the building block of quartz, amethyst, opal, agate, and most ordinary sand. Silicate minerals (compounds combining silicon, oxygen, and other elements) account for the majority of the rocks geologists classify as igneous and metamorphic. Outside the crust, silicon shows up everywhere in modern industry: it is the substrate of most semiconductor chips, the base of glass and ceramics, and a major ingredient in concrete and brick.
3. Aluminum (Al)
Aluminum is the most abundant metal in the Earth's crust, accounting for around 8.23% by weight. It never occurs free in nature because it bonds too readily with oxygen and other elements; the metal humans actually use is refined from bauxite ore, a mixture rich in aluminum oxide and aluminum hydroxide. Common aluminum compounds in the crust include feldspars (the most plentiful rock-forming minerals on the planet), micas, and alum salts such as potassium aluminum sulfate. Bauxite mining is concentrated in tropical countries where the weathering needed to concentrate aluminum oxide happens fastest.
4. Iron (Fe)
Iron makes up about 5.63% of the crust, but it dominates everything below the crust as well: the planet's outer and inner core are roughly 85% iron, and that iron is what generates Earth's magnetic field. The element gave its name to the Iron Age, the archaeological period that began in the eastern Mediterranean around 1200 BCE when smelted iron tools and weapons replaced bronze. Modern industry still runs on iron in the form of steel (an iron-carbon alloy used in everything from skyscraper frames to surgical instruments). In biology, iron is the central atom in hemoglobin, the protein that carries oxygen through human blood, and is essential to nearly every form of life on Earth.
5. Calcium (Ca)
Calcium accounts for about 4.15% of the Earth's crust. Calcium carbonate (CaCO₃) is the compound most readers will recognize from school chemistry: it is the main component of marble, chalk, limestone, and the shells of marine organisms such as oysters and corals, and it is the structural mineral in human bones and teeth. Calcium also forms gypsum (CaSO₄·2H₂O), the soft mineral processed into drywall and plaster of Paris, and it is essential in cement chemistry as the active ingredient in lime.
6. Sodium (Na)
Sodium accounts for about 2.36% of the Earth's crust and is one of the most familiar elements in the human diet, mostly in the form of sodium chloride (table salt). The pure metal is highly reactive and never occurs free in nature; cut a piece of sodium and expose it to air, and it tarnishes within seconds. Because sodium ions dissolve readily in water, the oceans hold an enormous reservoir of dissolved sodium, and salt flats such as the Bonneville Salt Flats in Utah and the Salar de Uyuni in Bolivia mark the locations where ancient saltwater bodies evaporated. In animals, sodium ions help regulate fluid balance and are central to nerve signaling and muscle contraction.
7. Magnesium (Mg)
Magnesium makes up about 2.33% of the crust and, like aluminum and sodium, never occurs as a free element in nature. The most common magnesium-bearing minerals include dolomite (calcium magnesium carbonate) and magnesite (magnesium carbonate). Outside geology, magnesium is the central atom of the chlorophyll molecule, the green pigment that drives photosynthesis in plants and most algae. The metal itself is unusually light, which is why aluminum-magnesium alloys are common in aircraft frames and high-performance bicycles.
8. Potassium (K)
Potassium accounts for around 2.09% of the crust and is, like the other alkali metals on this list, too reactive to occur in pure form in nature. The pure metal will ignite on contact with water. The most common natural sources are potash (potassium-bearing salts left behind by ancient evaporated seas) and minerals such as carnallite, sylvite, and polyhalite. Potassium chloride (KCl) is the foundation of the modern fertilizer industry; potassium carbonate (K₂CO₃) is used in soap-making and certain types of glass.
9. Titanium (Ti)
Titanium accounts for about 0.565% of the crust, and although that is far below the top eight, it is still significantly more abundant than any of the rare-earth elements that follow. The metal is found in rutile, ilmenite, and sphene, with most commercial production coming from rutile and ilmenite. Titanium has the highest strength-to-weight ratio of any metallic element, which is why it is used in aircraft, spacecraft, and military hardware. The same property, combined with its high biocompatibility, makes it the standard material for replacement hips, knees, and dental implants.
10. Hydrogen (H)
Hydrogen is the most abundant element in the universe by a large margin, but only the tenth most abundant in the Earth's crust at about 0.140% by weight. The reason for the gap is that hydrogen is a gas at Earth's surface temperatures and pressures, and most free hydrogen escaped Earth's gravity well during the planet's formation. The hydrogen that remains is locked into compounds: water (H₂O) accounts for the largest share, with the rest distributed across hydrocarbons, ammonia, hydrogen peroxide, sugars, and the bulk of organic molecules in living organisms. Industrially, hydrogen is the lightest gas and is increasingly produced as a clean fuel source, with hydrogen fuel cells now powering buses, trucks, and a growing range of vehicles.
The abundance gap between oxygen and the trace elements is vast. Oxygen at 461,000 ppm is more than 600 million times more abundant than rhenium at 0.0007 ppm, the rarest element in the table that follows. Even within the first ten, the gap between hydrogen at 1,400 ppm and phosphorus at 1,050 ppm is large, and abundance falls off quickly from there. The full ranking of 78 elements is below.
The Most Abundant Elements In The Earth's Crust
| Rank | Element | Symbol | Abundance in crust (ppm) |
|---|---|---|---|
| 1 | Oxygen | O | 461,000 |
| 2 | Silicon | Si | 282,000 |
| 3 | Aluminum | Al | 82,300 |
| 4 | Iron | Fe | 56,300 |
| 5 | Calcium | Ca | 41,500 |
| 6 | Sodium | Na | 23,600 |
| 7 | Magnesium | Mg | 23,300 |
| 8 | Potassium | K | 20,900 |
| 9 | Titanium | Ti | 5,650 |
| 10 | Hydrogen | H | 1,400 |
| 11 | Phosphorus | P | 1,050 |
| 12 | Manganese | Mn | 950 |
| 13 | Fluorine | F | 585 |
| 14 | Barium | Ba | 425 |
| 15 | Strontium | Sr | 370 |
| 16 | Sulfur | S | 350 |
| 17 | Carbon | C | 200 |
| 18 | Zirconium | Zr | 165 |
| 19 | Chlorine | Cl | 145 |
| 20 | Vanadium | V | 120 |
| 21 | Chromium | Cr | 102 |
| 22 | Rubidium | Rb | 90 |
| 23 | Nickel | Ni | 84 |
| 24 | Zinc | Zn | 70 |
| 25 | Cerium | Ce | 66.5 |
| 26 | Copper | Cu | 60 |
| 27 | Neodymium | Nd | 41.5 |
| 28 | Lanthanum | La | 39 |
| 29 | Yttrium | Y | 33 |
| 30 | Cobalt | Co | 25 |
| 31 | Scandium | Sc | 22 |
| 32 | Lithium | Li | 20 |
| 33 | Niobium | Nb | 20 |
| 34 | Nitrogen | N | 19 |
| 35 | Gallium | Ga | 19 |
| 36 | Lead | Pb | 14 |
| 37 | Boron | B | 10 |
| 38 | Thorium | Th | 9.6 |
| 39 | Praseodymium | Pr | 9.2 |
| 40 | Samarium | Sm | 7.05 |
| 41 | Gadolinium | Gd | 6.2 |
| 42 | Dysprosium | Dy | 5.2 |
| 43 | Erbium | Er | 3.5 |
| 44 | Ytterbium | Yb | 3.2 |
| 45 | Hafnium | Hf | 3.0 |
| 46 | Caesium | Cs | 3.0 |
| 47 | Beryllium | Be | 2.8 |
| 48 | Uranium | U | 2.7 |
| 49 | Bromine | Br | 2.4 |
| 50 | Tin | Sn | 2.3 |
| 51 | Europium | Eu | 2.0 |
| 52 | Tantalum | Ta | 2.0 |
| 53 | Arsenic | As | 1.8 |
| 54 | Germanium | Ge | 1.5 |
| 55 | Holmium | Ho | 1.3 |
| 56 | Tungsten | W | 1.25 |
| 57 | Molybdenum | Mo | 1.2 |
| 58 | Terbium | Tb | 1.2 |
| 59 | Thallium | Tl | 0.850 |
| 60 | Lutetium | Lu | 0.8 |
| 61 | Thulium | Tm | 0.52 |
| 62 | Iodine | I | 0.450 |
| 63 | Indium | In | 0.250 |
| 64 | Antimony | Sb | 0.2 |
| 65 | Cadmium | Cd | 0.15 |
| 66 | Mercury | Hg | 0.085 |
| 67 | Silver | Ag | 0.075 |
| 68 | Selenium | Se | 0.05 |
| 69 | Palladium | Pd | 0.015 |
| 70 | Bismuth | Bi | 0.0085 |
| 71 | Platinum | Pt | 0.005 |
| 72 | Gold | Au | 0.004 |
| 73 | Osmium | Os | 0.0015 |
| 74 | Tellurium | Te | 0.001 |
| 75 | Ruthenium | Ru | 0.001 |
| 76 | Iridium | Ir | 0.001 |
| 77 | Rhodium | Rh | 0.001 |
| 78 | Rhenium | Re | 0.0007 |