Why Does Ice Float?
Almost every other substance on Earth gets denser when it freezes. Water does the opposite. Solid ice is roughly 8 to 9% less dense than the liquid water it forms from, which is why it floats on the surface of lakes, oceans, and a glass of water rather than sinking to the bottom. The behavior is so unusual that it is sometimes called water's density anomaly, and the explanation comes down to one specific feature of water: the way water molecules attach to each other through hydrogen bonds, and the open three-dimensional lattice those bonds force water into when it freezes. The fact that ice floats is one of the more consequential chemical accidents in the history of life on Earth.
Most Solids Sink in Their Own Liquids
The general rule for materials is that solids are denser than their corresponding liquids. When you melt iron, copper, lead, or wax, the molten liquid is less dense than the solid, and any unmelted chunk dropped into the melt sinks. The reason is straightforward: in the liquid state, particles have enough thermal energy to break out of the regular geometric arrangement they hold in the solid, and most of the time the disordered liquid arrangement allows molecules to pack slightly more loosely than the regular solid arrangement. Less compact arrangement means lower density, so the liquid is the lighter of the two.
Water breaks the rule, and so do a small number of other substances. Silicon, germanium, gallium, bismuth, and a few alloys also expand when they freeze, for similar reasons related to crystal geometry. Water is the most familiar example because it is the only one any of us encounter at room temperature in both phases.
The Water Molecule and Hydrogen Bonds
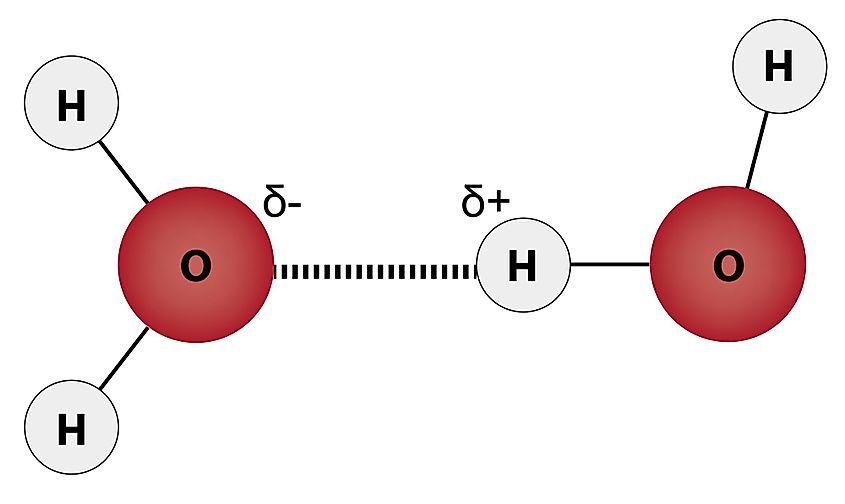
A water molecule is one oxygen atom and two hydrogen atoms held together by covalent bonds, in which each pair of atoms shares a pair of electrons. The molecule is not linear. The two hydrogen atoms sit on the same side of the oxygen at an angle of about 104.5° between them, giving the molecule a bent or boomerang shape. The oxygen atom is more electronegative than the hydrogen atoms, meaning it pulls the shared electrons more strongly toward itself. The result is that the oxygen end of the molecule carries a slight negative charge and the two hydrogen ends carry slight positive charges. Water is therefore polar, with a definite negative pole and two positive poles.
Polar molecules attract each other electrostatically, with positive ends drawn to negative ends on neighboring molecules. In water, this attraction is strong enough and specific enough to count as a distinct kind of bond, called a hydrogen bond. A hydrogen bond connects a slightly positive hydrogen atom on one water molecule to the slightly negative oxygen atom on a neighboring water molecule. Each water molecule can form up to four hydrogen bonds at once, two through its own hydrogens reaching out to neighboring oxygens, and two through its oxygen accepting hydrogens from other water molecules. Hydrogen bonds are about 5% to 10% as strong as the covalent bonds inside the molecule, but they are strong enough to dominate water's macroscopic behavior.
The Open Lattice of Ice
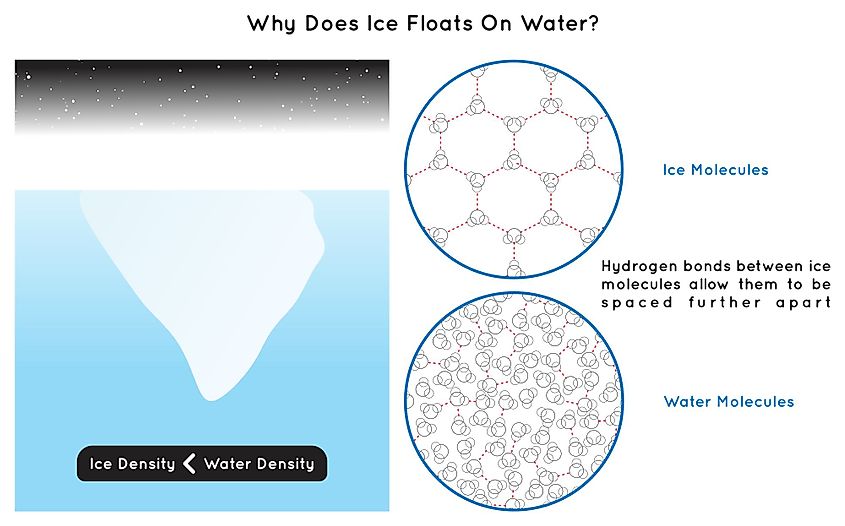
In liquid water, hydrogen bonds are continuously forming and breaking on a picosecond timescale. At any given instant, each water molecule has fewer than four hydrogen-bonded neighbors on average, and the molecules can slide past each other and pack into whatever arrangement minimizes their energy at that moment. The arrangement is dense and disordered.
When liquid water cools below 0°C and freezes, the hydrogen bonds lock into place. Each water molecule becomes hydrogen-bonded to exactly four neighbors arranged in a tetrahedral geometry around it: two oxygens above and two below, each at the corner of a regular tetrahedron. The repeating unit of this arrangement, called Ice Ih, is a hexagonal crystal lattice, which is why snowflakes are six-sided. The catch is that the tetrahedral arrangement of bonds is not a compact way to pack molecules. There is significant empty space inside the hexagonal rings of the lattice. Liquid water can crowd molecules into that space; solid ice cannot. The fixed geometry of the lattice forces ice to occupy more volume per molecule than the same number of molecules occupied as a liquid, and ice ends up roughly 8 to 9% less dense.
It is worth noting that Ice Ih is only one of the many forms ice can take. Researchers have now identified roughly 20 distinct crystalline phases of ice, most of them stable only under extreme pressure or extreme cold. Some of these high-pressure ices are denser than liquid water and would sink in it. Ice VII, for example, exists at pressures above 2 gigapascals (about 20,000 atmospheres) and has been detected as natural inclusions inside diamonds brought up from deep in the mantle. The ice that forms in a freezer or on the surface of a pond is exclusively the low-pressure form, Ice Ih, which is why ordinary ice always floats.
Why Water Reaches Its Maximum Density at 4°C
Water has an unusual temperature-density relationship even before it freezes. As pure water cools from room temperature, it becomes denser, as expected, but only down to a temperature of 3.98°C (often rounded to 4°C). Below that point, water keeps cooling but begins to expand again, becoming less dense rather than more. This anomalous expansion continues all the way to 0°C, at which point the water freezes and expands sharply.
The reason is that the same hydrogen-bonded tetrahedral arrangement that builds Ice Ih starts to assert itself in the liquid as the temperature falls. As water approaches the freezing point, more and more of the molecules at any given instant are arranged in transient hydrogen-bonded clusters that resemble the open lattice of ice. The clusters take up more volume per molecule than disordered liquid water does, and they grow more numerous as temperature drops. The two effects compete: ordinary thermal contraction that pulls molecules closer as they cool, versus the expansion driven by the growing fraction of ice-like clusters. Above 4°C, contraction wins. Below 4°C, expansion wins. The crossover point is the maximum-density temperature.
This sounds technical, but it has direct ecological consequences. In a freshwater lake in winter, the water at the surface cools first. As it cools, it gets denser and sinks, while warmer water from below rises to take its place. This convective mixing continues only down to 4°C. Once the surface reaches 4°C, any further cooling makes the surface water lighter rather than heavier, so it stops sinking and stays at the top. The cold layer cools further, eventually reaching 0°C, and then ice begins to form on the surface, where it stays. The deeper water in the lake remains at roughly 4°C all winter, allowing fish, amphibians, and aquatic invertebrates to overwinter in liquid water beneath the ice.
The 9% Above the Surface

The proportion of ice that floats above the waterline follows directly from Archimedes' principle. A floating object displaces a volume of liquid equal in weight to the object itself. Pure ice has a density of about 917 kg/m³, and pure water near 0°C has a density of about 1,000 kg/m³, so the ratio is 917/1000 = 0.917. About 91.7% of an ice cube floating in fresh water sits below the surface, and about 8.3% pokes up above it. In seawater, which is denser than fresh water at around 1,025 kg/m³, the submerged fraction drops slightly to about 89.5%. The figure of "an iceberg's tip is one-tenth of the iceberg" is a reasonable rough rule for either case.
The same expansion that makes ice float also explains why pipes burst when water freezes inside them, why potholes form in roads after wet winters, and why glaciers carve valleys: water seeps into cracks, freezes, and exerts pressure as it expands. The pressure generated by water freezing in a fully sealed container can exceed 30,000 pounds per square inch, easily enough to split steel.
Why This Matters for Life on Earth
The fact that ice floats has shaped the conditions for life. The most direct consequence is the one already mentioned for lakes: aquatic life can survive winter beneath an insulating layer of ice rather than being killed off when the entire water column freezes solid. The same principle operates at much larger scales. The polar oceans freeze from the surface down, building floating sea ice that, in turn, supports an entire ecosystem (sea ice algae, copepods, krill, polar cod, seals, polar bears) above and below the ice. If ice were denser than water, the polar oceans would freeze from the bottom up, slowly accumulating a permanent layer of ice on the seafloor that the limited summer melt at the surface could never reach. The deep oceans would gradually solidify, the planet's climate would behave very differently, and any biosphere that managed to develop would look almost nothing like the one we have.
The implications extend beyond Earth. The search for life on other worlds in our solar system has increasingly focused on subsurface oceans, particularly on Jupiter's moon Europa and Saturn's moon Enceladus, both of which appear to have liquid water beneath thick ice shells. Those ice shells exist in part because the water-ice density inversion produces a stable floating crust over a liquid interior, the same way it does on a frozen lake. Without the anomaly, ocean worlds with active subsurface hydrology would be far less likely.
The fact that ice floats is, in other words, not a curiosity. It is one of the structural features of how water behaves, and a great deal else depends on it.