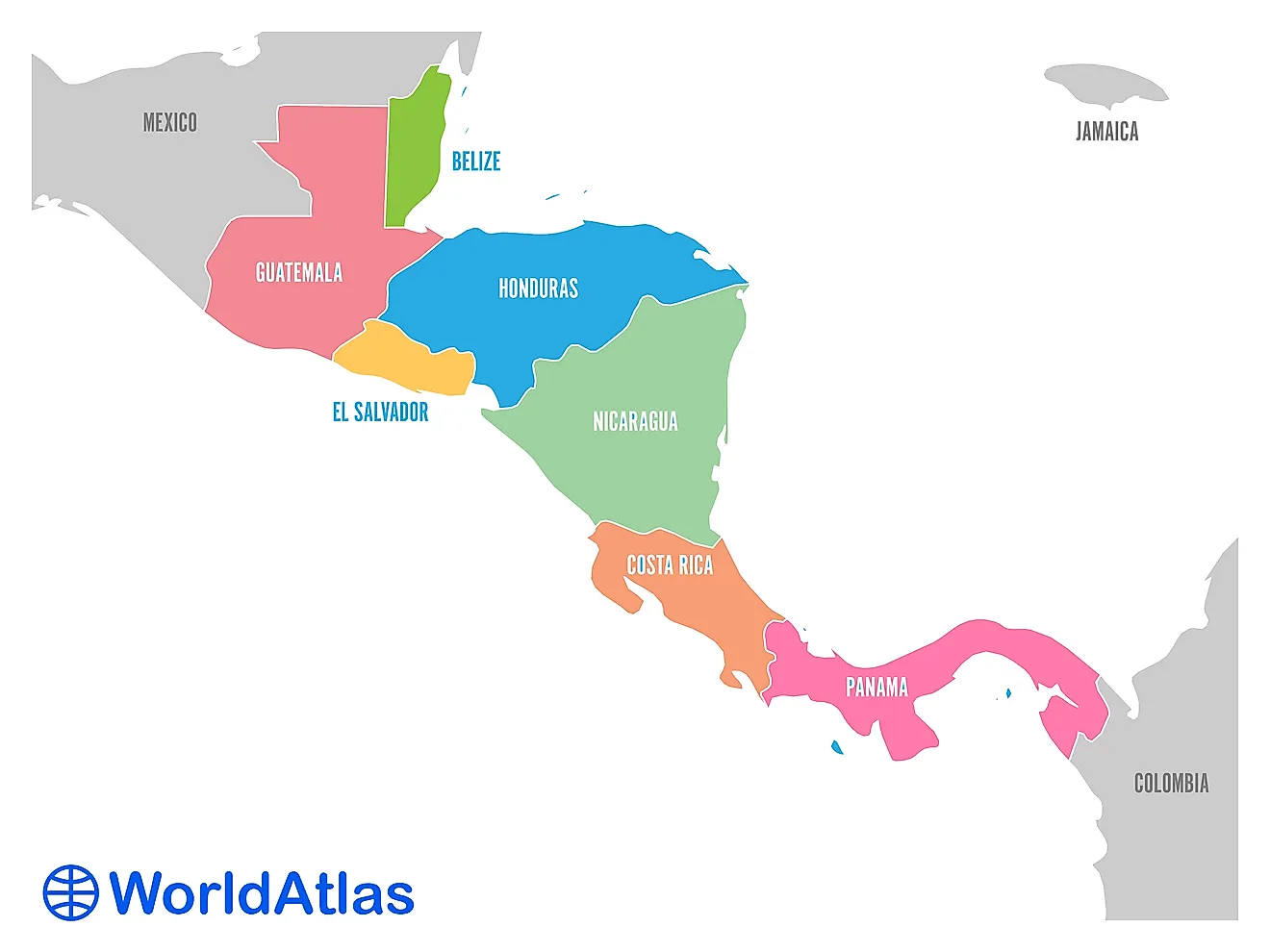
What Is Antimony?

Antimony is a shiny gray metalloid which exists in nature as sulfide mineral stibnite. The atomic number of antimony is 51, and its chemical symbol is Sb, which is derived from the Latin term "stibium." Antimony compounds, commonly known by the Arabic name ‘’kohl,’’ have been used as cosmetics and medicines since ancient times. Metallic antimony was mistakenly recognized as lead when first discovered. Antimony was first isolated and described by Italian metallurgist Vannoccio Biringuccio in 1540.
When Was Antimony Discovered?
Antimony trisulfide was discovered as early as 3100 BC in predynastic Egypt, and was used in cosmetics, such as eye makeup. An artifact made of antimony, which dates back to 3000 BC, was discovered in Telloh Chaldea, and an anitomy-plated object dating between 2500 BC to 2200 BC was discovered in Egypt. The first procedure for extracting antimony was written by Vannoccio Biringuccio in 1540 in his book the De la pirotechnia. The first natural occurrence of antimony was described by Swedish scientist Anton Von Swab in 1783, and the first sample of pure antimony was mined in the Sala silver mines in Sala, Sweden.
What Are the Characteristics of Antimony?
Antimony is a chemical element from group 15 of the periodic table, called a pnicyogen, and has an electronegativity of approximately 2.05. Antimony is significantly more electronegative than arsenic or tellurium. Antimony is relatively stable at room temperature, but undergoes oxidation to form antimony trioxide when heated. Antimony has a Mohs scale hardness of three, which is too soft to build hard objects. In fact, Guizhou, China used antimony to make coins in 1931, but discontinued minting them due to their poor durability. Antimony is resistant to acid attacks.
There are four known antimony allotropes: stable metallic antimony; and 3 meta-stable forms (yellow, black and explosive). Elemental antimony is a delicate and shiny silvery-white metalloid. When cooled, antimony always crystallizes into an isomorphic trigonal cell, with a gray arsenic allotrope.
Antimony trichloride creates a rare volatile form of antimony each time it undergoes electrolysis.
An exothermic reaction occurs when antimony is scratched, and when rubbed in a mortar with a pestle, a strong explosion occurs. At 100 degrees Celsius, this compound transforms into a stable form. The most unstable structure of this compound is the yellow allotrope, which can only be produced by the oxidation process of stibine at a temperature of negative ninety degrees Celsius.
Where Is Antimony Found?
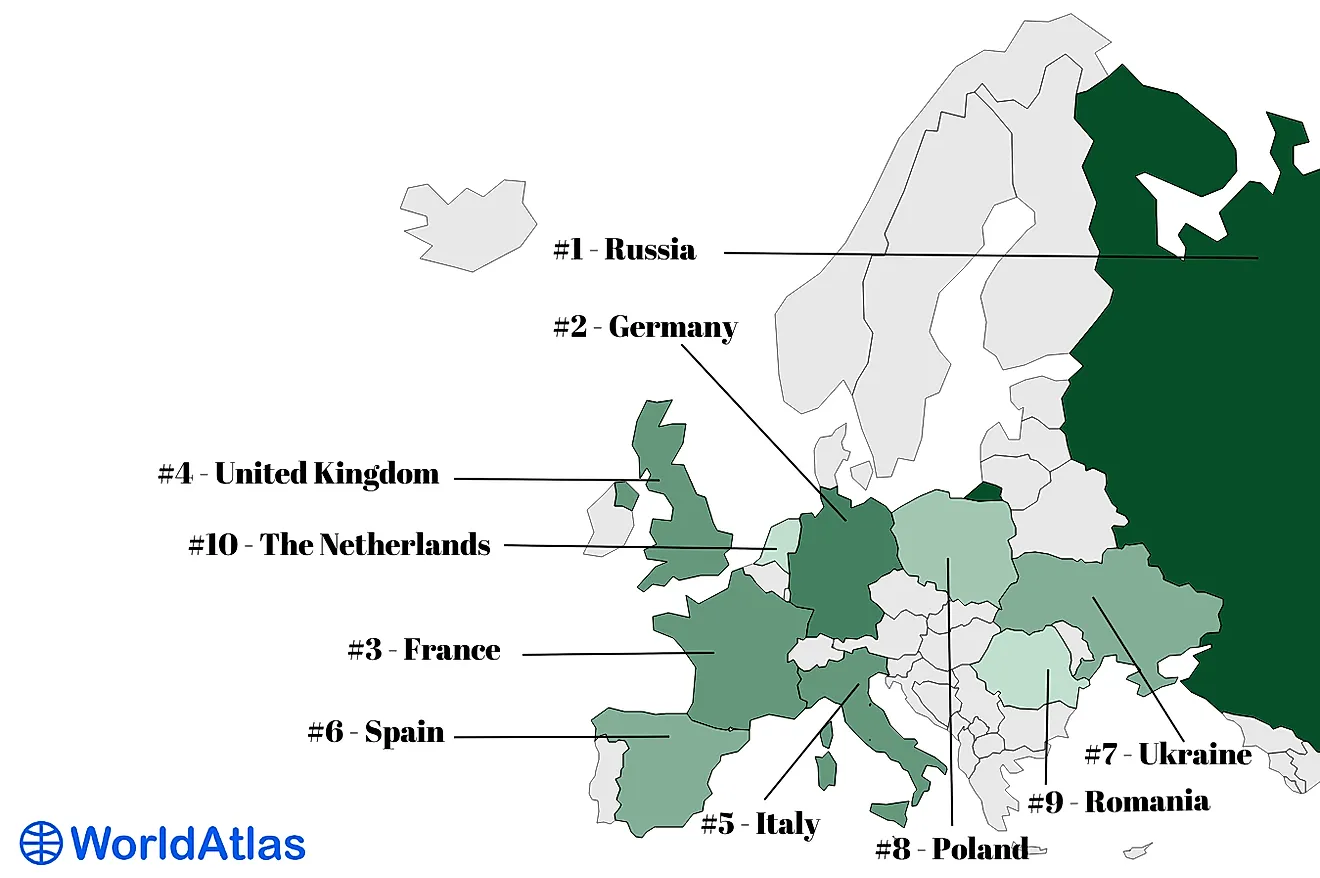
The amount of antimony in the crust of the Earth is between 0.2 to 0.5 parts per million (ppm). Although the compound is scarce, it is present in more than a hundred minerals. A pure sample of antimony can be extracted, but most of the time antimony is present in sulfide stibnite, which is the principal ore material.
How Many Isotopes Does Antimony Have?
Antimony has only two stable isotopes: 121Sb, which has an abundance of 57.365%; and 123Sb, which has a wealth of 42.645%. Antimony has about thirty five radioisotopes, and Sb has a half-life of two years and nine months. Isotopes that are lighter than Sb tend to decay by the β+ decay (positron emission), while heavier ones decay by β− decay (electron emission).