What Are The Properties Of Matter?

- There are six major physical properties. In order for us to measure or observe them, we do not need to change the composition of the substance. The six physical properties are color, density, volume, mass, boiling point, and melting point.
- Chemical properties are those which we can measure only by changing the chemical structure of the substance we are measuring.
- The four most important chemical properties are the heat of combustion, chemical stability, flammability, and the preferred oxidation state.
All matter has certain properties that define it. We can usually measure those properties, and determine the size, weight, or the composition of a matter. For example, we can determine that wood is mostly brown, thereby detecting its property of color. Several properties are considered to be essential, and those are most often divided into physical and chemical properties.
We also differentiate between extensive and intensive properties of matter. Physical properties are those that can be measured or observed without changing the chemical composition of a matter. Things like volume and color fall under this category.
On the other hand, chemical properties can only be measured or observed when we change the chemical identity of a substance. Extensive and intensive properties are just physical properties but divided into two specific groups. Extensive properties, such as volume and mass, depending on the amount of substance we are measuring, while intensive properties, such as color and density, do not.
Physical Properties
There are six major physical properties. In order for us to measure or observe them, we do not need to change the composition of the substance. The six physical properties are color, density, volume, mass, boiling point, and melting point. Out of those six, four are intensive, while the other two are extensive.
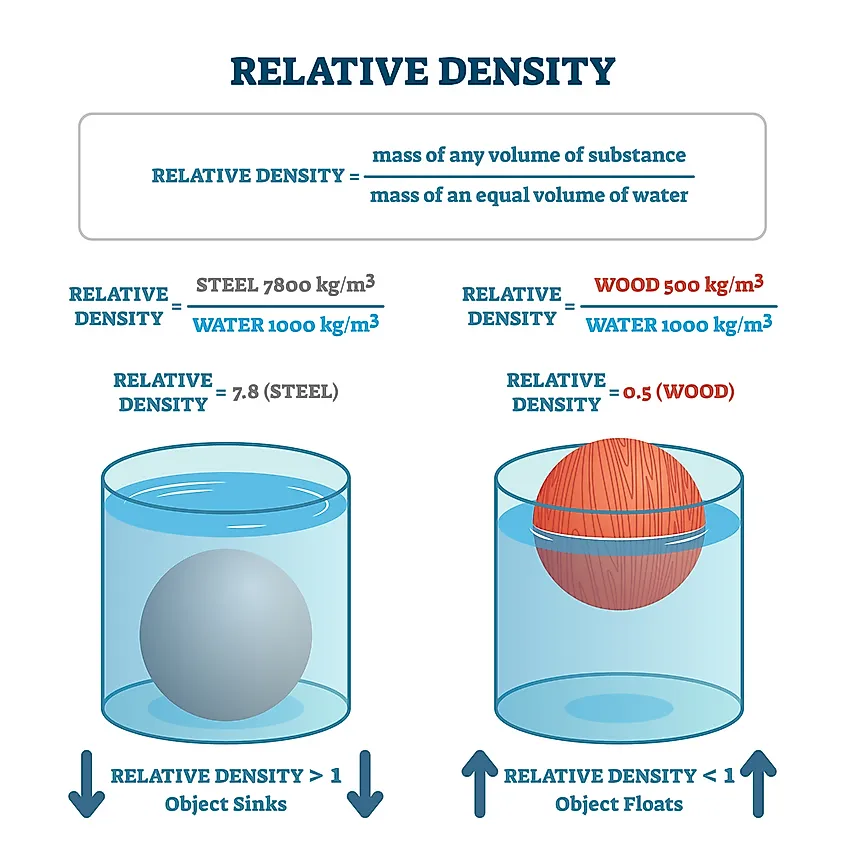
The extensive physical properties are volume and mass. Their value depends on the amount of matter we are measuring. The intensive properties of matter are color, density, boiling point, and melting point. These are permanent; they do not change depending on the amount of matter. It makes sense, no matter how much of a specific matter you have, its color won’t change.
There are other properties we can mention here, but those fall under these six, which are considered the most important. Other properties of matter that deserve mention are refractive index, temperature, and hardness, for example. These are all intensive properties, often described as bulk properties. They do not depend on the size of the system we are measuring, or the amount of material that can be found in that system.
Chemical Properties
We already mentioned that chemical properties are those which we can measure only by changing the chemical structure of the substance we are measuring. This means that these properties measure how matter behaves when put inside of a chemical reaction. These properties can become evident only during such a reaction. The four most important chemical properties are the heat of combustion, chemical stability, flammability, and the preferred oxidation state.
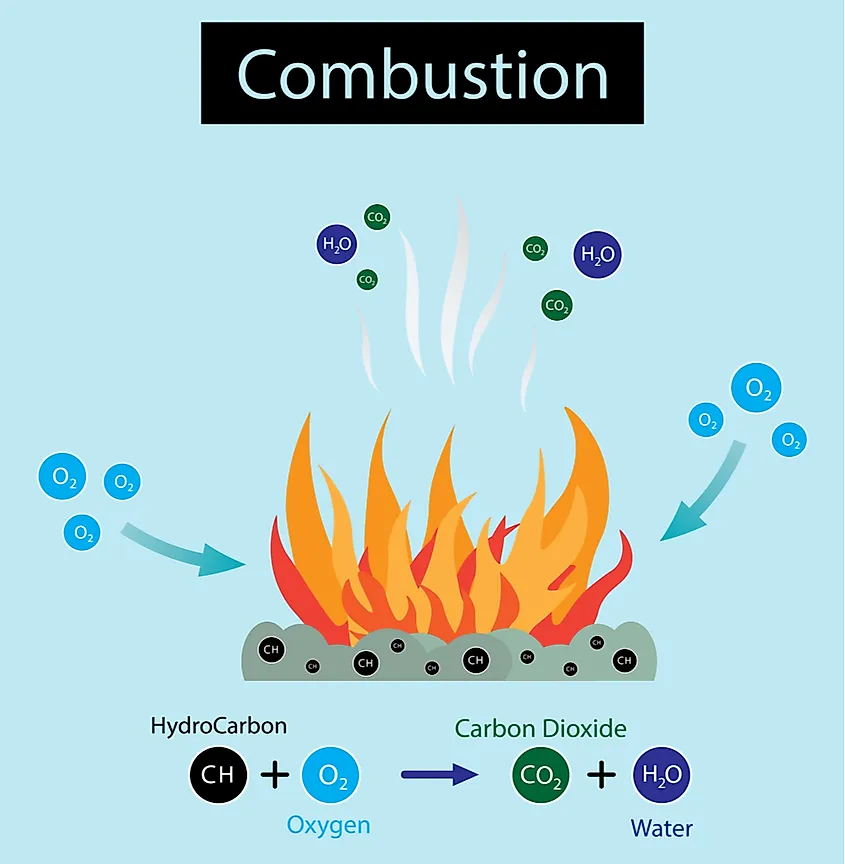
The heat of combustion is the property of the energy that gets released when matter completely combusts through the use of oxygen. This property measures the amount of energy that gets released during such a reaction. Chemical stability is the property that determines if a matter will react when placed in a specific environment. It is mostly observed by placing a compound in water or just leaving it outside to see how it reacts with air. These reactions are called hydrolysis and oxidation.
The next chemical property is flammability, which measures the ability to combust. Through flammability, we can see if the matter will burn once we expose it to fire. Burning is a chemical reaction, and by placing matter under it, we can determine how flammable it is, and how quickly it will burn. Finally, the preferred oxidation state determines the level of oxidation of a given compound that requires the least amount of energy. Oxidation state, in general, is the degree of oxidation (the loss of electrons) of a compound, and the preferred one is the one that needs the lowest amount of energy.