Are We Made Of Stardust?
The astronomer Carl Sagan once famously said that “we are made of starstuff.” Interestingly, this is actually an accurate statement about the universe. Not only are humans made of starstuff, but everything we see around us is, along with everything in the solar system. The elements that make up our bodies and all the planets were cooked in the cores of massive stars.
Where Elements Come From
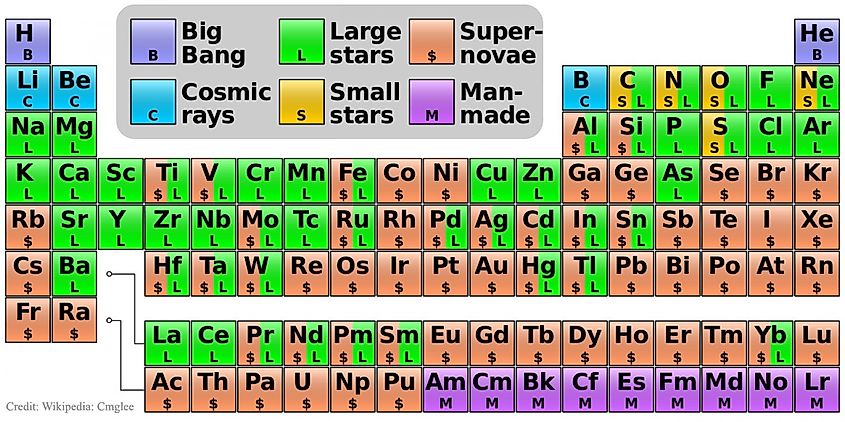
When the universe began 13.8 billion years ago in the Big Bang, conditions were far too extreme for ordinary matter to form. Temperatures were too high for even individual quarks to come together to form neutrons and protons. However, within the first few minutes of existence, the universe cooled enough for quarks to form the first protons and neutrons. However, it took another 380,000 years for things to cool enough for the first atoms to form. As soon as this occurred, the universe became transparent and light began making its journey across the cosmos. The first atoms were almost entirely hydrogen, with smaller amounts of helium and lithium as well. Despite most atoms being hydrogen, we know our universe now contains an abundance of other elements. There are a total of 118 elements, 92 of which are found in nature. to be exact. Hydrogen is the simplest and most abundant element in the universe, and all of the other elements originated from hydrogen. In order to turn hydrogen into heavier elements, stars needed to form. Stars are almost entirely composed of hydrogen, yet within their cores, hydrogen is actively fused into heavier elements such as carbon and oxygen through the process of nuclear fusion. Heavier stars are able to continue the fusion process further down the periodic table than low mass stars. The most massive stars are able to eventually create iron within their cores. Once this occurs, the star is doomed. The fusion of iron requires more energy than it releases, and thus, iron begins to absorb the star’s energy. As this occurs, the star’s immense gravity takes over and the star collapses. The core is crushed to such an extent that the repulsive forces between atoms are overcome. Protons and electrons merge together to form neutrons. Eventually, the core is almost entirely composed of neutrons and becomes what’s called a neutron star.
Supernova
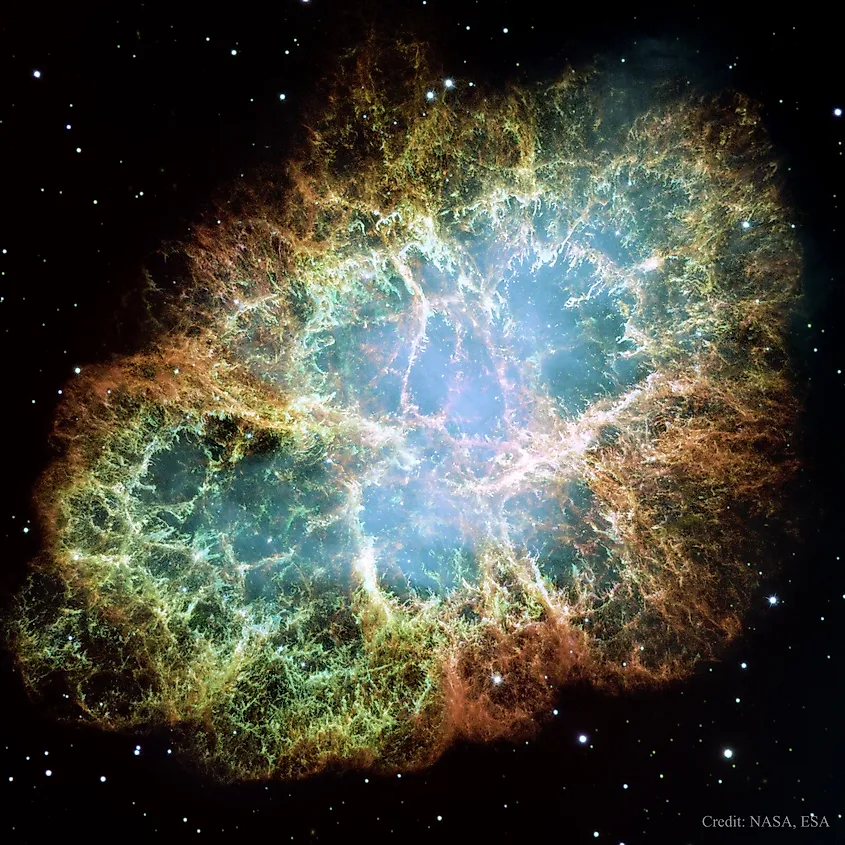
The rest of the star continues to collapse until it rebounds off the neutron star. The result is a gigantic explosion called a supernova. During a supernova, the amount of energy produced is so high that iron can be fused into even heavier elements, such as gold and silver. If star’s did not cease to exist one day, the elements they fuse would remain locked within their cores. In order for the very ingredients of planets and life to find their way into space, stars must explode. Our very existence is only made possible due to the laws of physics that govern stellar life cycles. If it were not for the stars, the universe would be nothing but a cloud of hydrogen. The atoms that compose nearly everything we see were once located within the cores of massive stars.