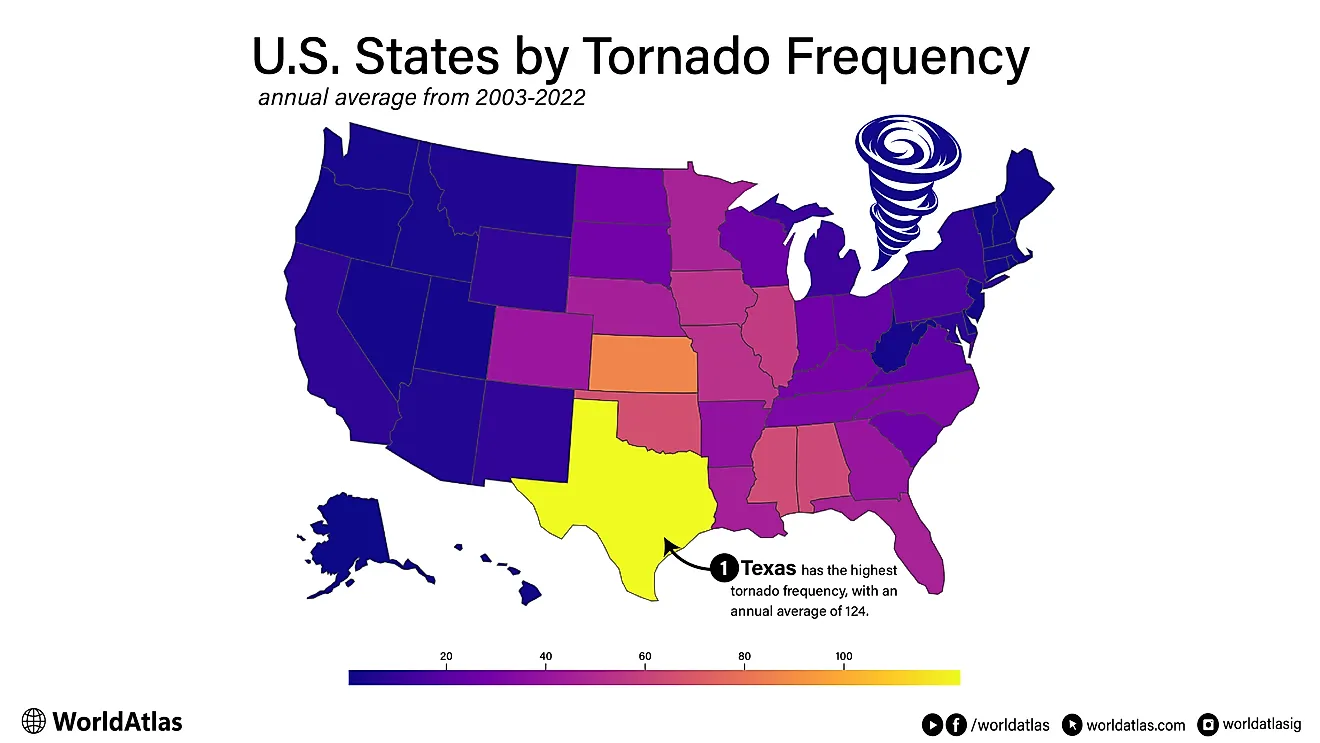
The Most Common Elements In The Universe
All of the visible matter we can see around us, from the Earth to the distant stars, is composed of various elements. The elements are organized into the periodic table containing 118 known elements. Of those 118 elements, 92 occur naturally, and the others have been created by humans through experiments or as byproducts of nuclear reactors. Of those 92 elements, which are the most common in the universe?
Where Elements Come From
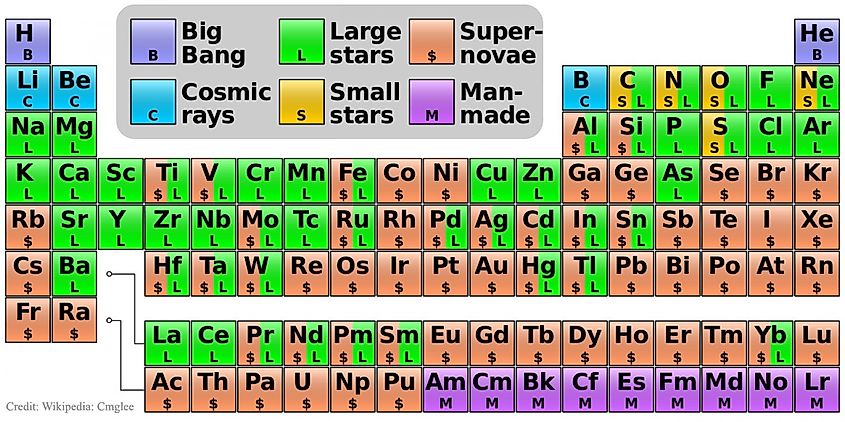
The universe originated from the Big Bang nearly 14 billion years ago. Immediately after, temperatures were so extreme that elements couldn't form; instead, it was a hot expanse of energy, electrons, and quarks. As it cooled, quarks grouped in threes to create protons and neutrons. The first element to form was hydrogen, consisting of a single proton, but without electrons, it wasn't a true element. Still, some protons fused, producing helium nuclei and small amounts of lithium. It took about 380,000 years for temperatures to drop enough for electrons to combine with nuclei, creating the initial elements. At this stage, only hydrogen, helium, and lithium existed naturally. Heavier elements are produced later in the cores of massive stars via nuclear fusion.
The Most Common Elements

As one might expect, the abundance of any given element is somewhat dependent on its simplicity. Hydrogen has only one proton and one electron, and thus it is the most abundant element in the universe. After hydrogen, helium is the most abundant element in the universe. In fact, hydrogen and helium combined make up more than 99% of all visible matter in the universe. Less than 1% of all that we can see is composed of everything heavier than helium. The ten most common elements in the universe are hydrogen, helium, oxygen, carbon, neon, iron, nitrogen, silicon, magnesium, and sulfur. It may seem strange that some lighter elements, such as lithium and beryllium, are not in the top ten. Despite some elements being lighter and simpler than those listed, they are not nearly as common. That’s because some elements are destroyed during the fusion process within stars, making them exceedingly rare in the cosmos. Furthermore, elements heavier than iron are so rare because stars can create them only for a short period of time. Fusing elements heavier than iron absorbs far more energy than it releases, and so when stars make iron in their cores, they reach the end of their lives. The most massive stars will explode in mighty supernovae, dispersing their newly formed elements into space. Those elements will one day be used in the formation of another generation of stars and planets.