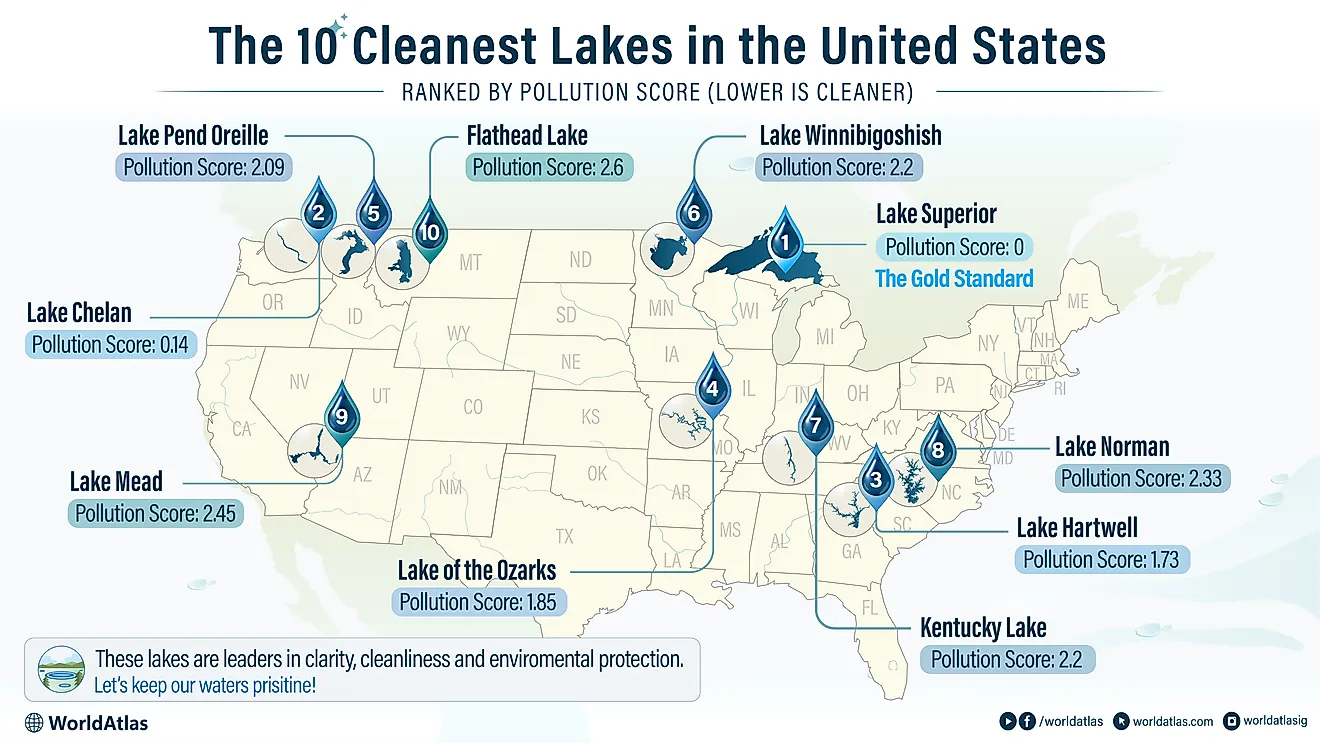
The Ocean Carbon Cycle
Carbon is the most essential tetravalent, non-metallic element that forms a fundamental component of all life on Planet Earth. After hydrogen, helium, and oxygen, carbon is the 4th most prevalent element in the universe by mass and the 15th most abundant element in the Earth's crust. Oceans are an extensive carbon reservoir that contains 36,000 gigatonnes of carbon, roughly 60 times more than the atmosphere. Also known as the Marine Carbon Cycle, the Oceanic Carbon Cycle refers to the processes by which carbon is continuously exchanged within the ocean as well as with the atmosphere. Through the Oceanic Carbon Cycle, the oceans play an essential role in mitigating global warming by absorbing excess carbon dioxide emissions. However, as anthropogenic climate change continues to impact ocean circulation, ecosystem dynamics, and the crucial biogeochemical cycles, the future fate of oceans as an important carbon sink is day-by-day becoming quite uncertain.
Oceanic Carbon Cycle
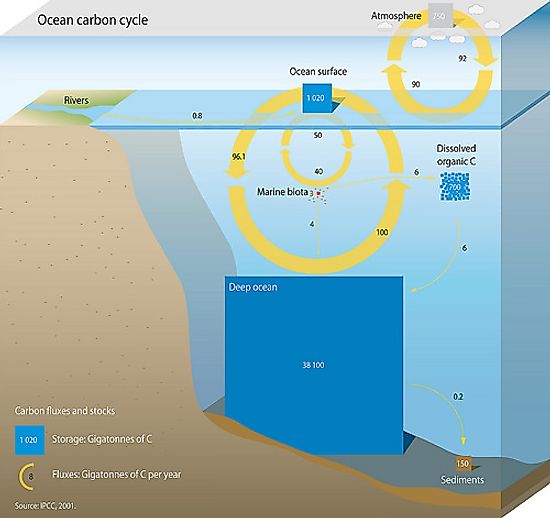
Due to the vast size of the oceans and comparatively rapid exchange of carbon dioxide with the atmosphere, the Oceanic Carbon Cycle plays a critical role in controlling atmospheric carbon dioxide concentration over the timescales of centuries or longer. Therefore, any slight changes in the natural components of this Marine Carbon Cycle will significantly affect the Earth’s climate system.
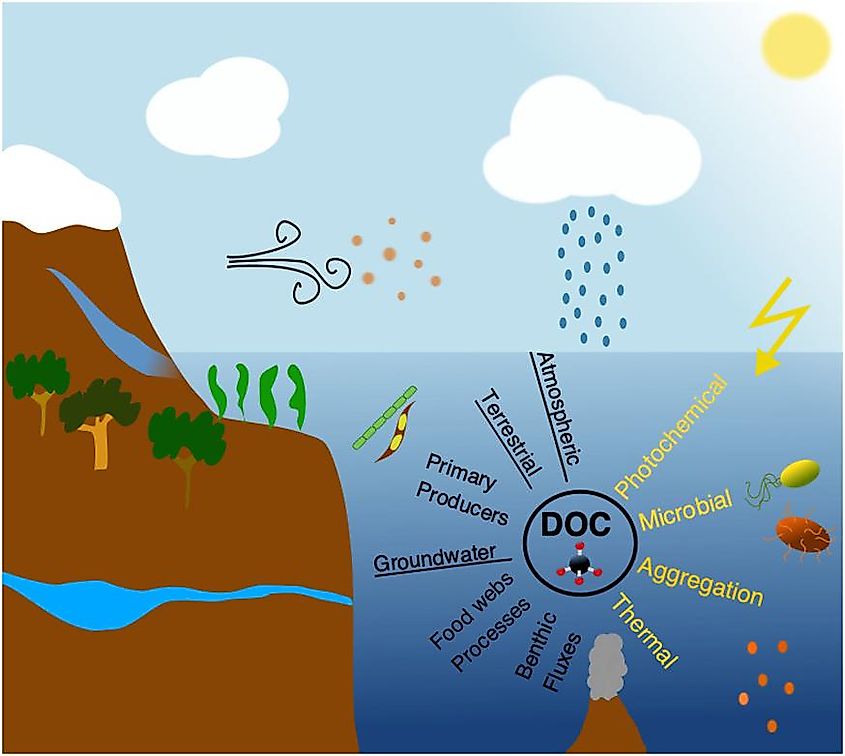
The Oceanic Carbon Cycle contains both inorganic and organic carbon depending on the composition. Inorganic carbon is found in simple compounds like carbon dioxide, carbonates, carbonic acid, and bicarbonate. Organic carbon forms the main component of organic compounds like proteins, carbohydrates, nucleic acids, and lipids. Moreover, the inorganic carbon in the oceans can further be classified into two types: Dissolved inorganic carbon (DIC) and Particulate inorganic carbon (PIC). Similarly, there are two forms of organic carbon in the oceans: Dissolved organic carbon and Particulate organic carbon.
Marine Carbon Pumps
In simple terms, the equilibrium carbon capacity of the carbon-dioxide system combined with the relative masses of the ocean and the atmosphere helps to determine the carbon partitioning between the ocean and the atmosphere. However, the Dissolved inorganic carbon shows a strong vertical gradient in the ocean, with low concentrations close to the surface and comparatively high concentrations at the depths. Ocean carbon pumps maintain these gradients of the Dissolved inorganic carbon and are therefore crucial in regulating carbon partitioning between the ocean and the atmosphere.
Solubility Pump
Solubility pump refers to the physicochemical process by which carbon is transported from the ocean's surface to its depths as dissolved inorganic carbon. This solubility pump is, in turn, driven by two oceanic processes: the solubility of carbon dioxide in water and the Global Ocean Conveyor Belt (Thermohaline Circulation). It is to be noted that carbon dioxide is more soluble in cold waters; therefore, at high latitudes where surface cooling occurs, this carbon-dioxide-laden water sinks into the deep ocean and becomes a part of the thermohaline circulation. At warmer equatorial latitudes, when this deepwater upwells, carbon dioxide is returned into the atmosphere.
Carbonate Pump
Often referred to as the “carbonate counter pump,” the carbonate pump begins with the marine organisms found at the ocean surface and which generates particulate inorganic carbon in the form of calcium carbonate. This calcium carbonate results in the formation of the hard body shells of these marine organisms. The shell formation, in turn, increases atmospheric carbon dioxide. The ecologically significant exclusively marine Coccolithophore phytoplankton group plays a vital role in the oceanic carbon cycle. These organisms which produce calcium carbonate shells are the carbonate pump’s essential contributors and help in the downward transport of calcium carbonate.
Biological Pump
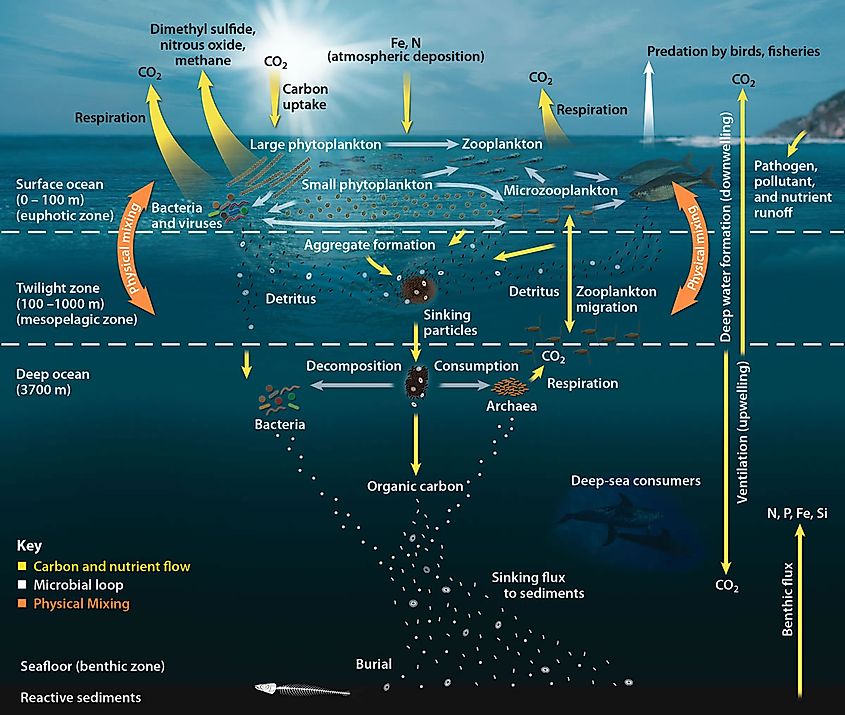
The marine biological carbon pump is at the heart of the Oceanic Carbon Cycle. This biologically mediated process involves marine microorganisms' photosynthetic uptake of atmospheric carbon dioxide, which results in carbon sequestration in the deep oceans away from the land and the atmosphere. The biological pump has different phases: the first involves the production of fixed carbon by the planktonic phototrophs through photosynthesis in the ocean's euphotic zone. Some planktons combine calcium and dissolved carbonates to form their protective calcium carbonate coating.
Once this carbon is fixed, the marine microorganisms either stay in the sunlight zone to be recycled or if they die, they sink to the ocean floor. This fixed carbon then undergoes decomposition by bacteria and remineralizing to be used again for primary production. Some particles which escape these processes are sequestered in the sediment and remain there for millennia. This sequestered carbon is eventually responsible for lowering atmospheric carbon dioxide concentrations.
It is estimated that the biological pump overall transfers approximately 10.2 gigatonnes of carbon yearly into the ocean's depths and, on average, a total of 1300 gigatonnes of carbon over 127 years. This means the carbon remains out of contact with the atmosphere for thousands of years. Moreover, without a biological pump in the oceans, the level of atmospheric carbon dioxide would be about 400 ppm higher than in the present times.
Inputs And Outputs Of The Ocean Carbon Cycle
There are numerous inputs to the marine carbon cycle, but on a net basis, the atmosphere and the rivers make the principal contributions. Rivers transport organic carbon to the oceans through different processes like weathering and erosion of minerals and carbonate rocks. Before the beginning of the Industrial Revolution, the atmospheric carbon dioxide concentration was roughly 280 ppm. During this period, the flow of carbon in and out of the atmosphere was closely balanced. However, over the years, the atmospheric carbon dioxide concentration has been rapidly increasing due to industrial activities. The exchange rate at which carbon dioxide is absorbed at the ocean surface from the atmosphere varies greatly. Moreover, the capacity of the oceans to take in carbon dioxide decreases with the rise in sea surface temperatures.
The marine carbon system’s primary outputs include the particulate organic matter and preservation of calcium carbonate. The climate of the Earth is moderated by the flow of carbon in and out of the lithosphere on time scales greater than 500,000 years. The rocks in the ocean depths are recycled back to the surface via plate tectonics to undergo weathering or subduction into the mantle. The carbon is then outgassed into the atmosphere by volcanic eruptions.
Impacts Of Climate Change On The Ocean Carbon Cycle
Human activities like fossil fuel burning, deforestation, rapid expansion of agricultural practices, and industrial activities have increased carbon dioxide release into the atmosphere. The ocean functions as a carbon sink for anthropogenic carbon dioxide. However, this excess carbon dioxide has decreased the pH levels of the Earth’s oceans. Ocean acidification also lowers carbonate saturation levels, significantly affecting the marine calcifying organisms that depend on calcium carbonate to build their outer shells and skeletons. Moreover, these marine organisms suffer from depressed metabolic rates, reduction in energy for essential functions, and low immune responses. The marine ecosystems that provide food, livelihood, and other ecosystem services are also significantly affected by ocean acidification.
As evident from the above discussion, the ocean carbon cycle plays a vital role in controlling the Earth’s climate. Any small changes in the components of the cycle can have disastrous consequences. Therefore, a thorough understanding is necessary to predict how future climate scenarios can affect the Ocean Carbon Cycle.