Carbon Cycle
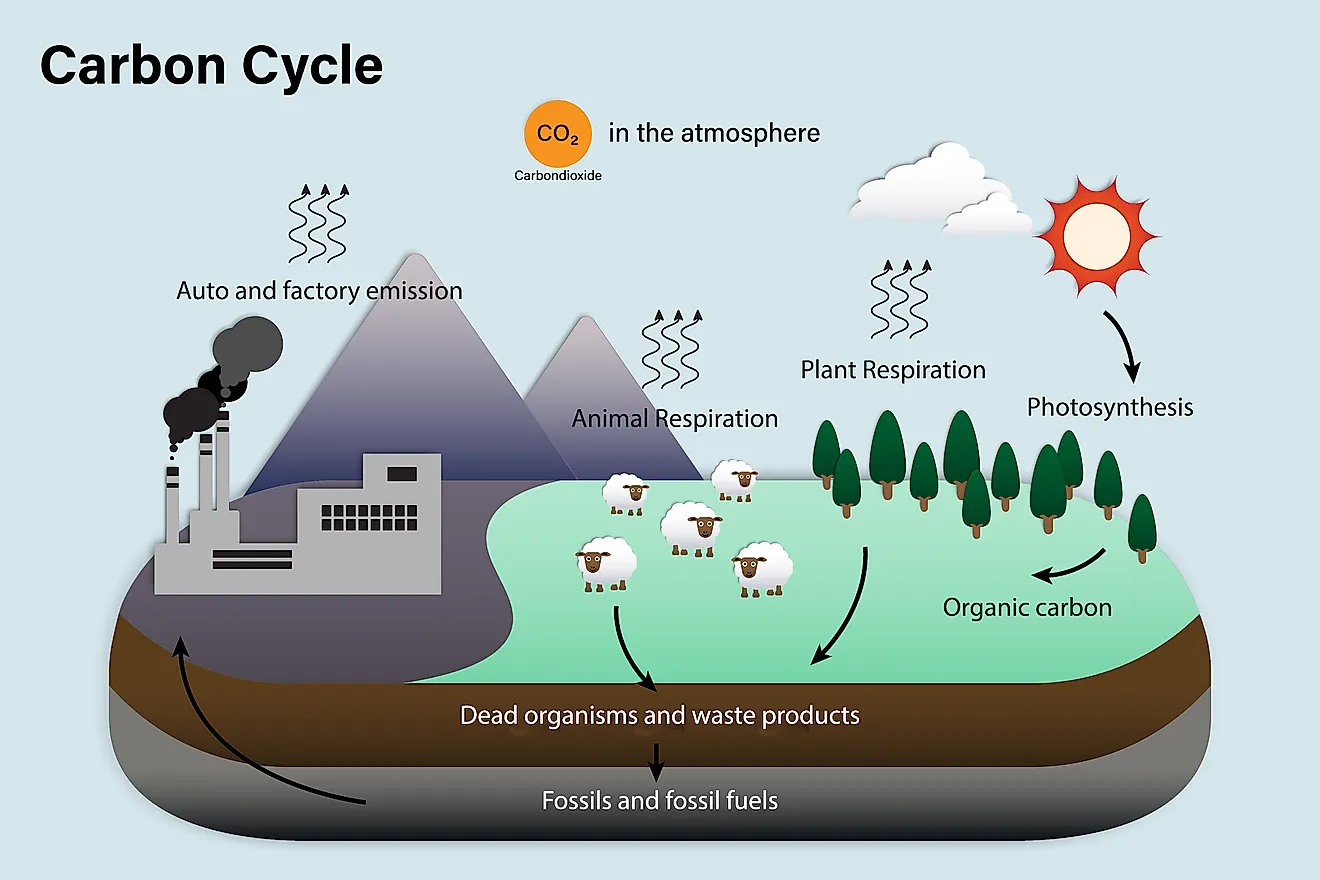
- 1,000 to 100,000 million metric tons of carbon move through the fast carbon cycle every year.
- The Carbon Cycle moves in two major ways, known as the fast cycle and slow cycle.
- Photosynthesis, consumption, respiration and decomposition are the major processes through which carbon moves within the carbon cycle.
Carbon is one of the many natural elements that can be found on, and in, Earth. It is one of the most abundant elements after hydrogen, helium, and oxygen, and is an integral part of all human, animal, and plant life. Carbon is especially important in biology because it can easily form multiple bonds in a variety of organic molecules. These bonds are strong, meaning they can form into rings and long connected chains, which are the backbone of most living cells.
These carbon bonds also hold a lot of energy, which can be stored up, or released if the bonds are broken. Because of this, carbon is an important and often-used source of fuel for living things.
Where Is Carbon Found?
Carbon can be found in all four spheres of the Earth. The lithosphere, or rock and earth aspect of the Earth, holds a large amount of carbon. In fact, it is in the top 15 elements that form the Earth’s crust. Carbon can also be found in the atmosphere, most commonly in the form of carbon dioxide, an organic compound composed of both carbon and oxygen. The hydrosphere, which includes all bodies of water on Earth, also contains carbon in carbon dioxide form, as does the biosphere, which is the term used for all organic life.
The Carbon Cycle
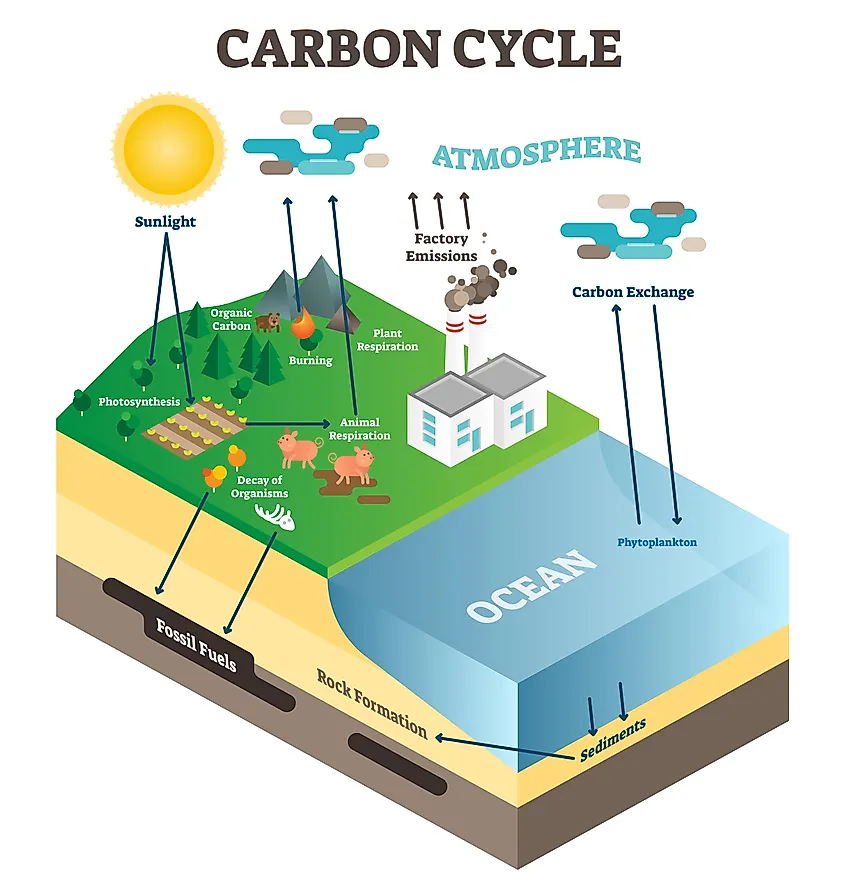
The carbon on Earth, and in the four earthly spheres, is constantly in motion. This exchange of carbon and carbon compounds is what is known as the Carbon Cycle. There are various processes, both natural and synthetic, that cause carbon to be exchanged from one sphere to another. Sometimes the carbon moves quickly within the Earth’s systems, and other times it can be retained in one place for many years, and thus is exchanged at a much slower rate. Because of this, two distinctions have been made. These are known as the Fast Cycle and the Slow Cycle.
The Fast Cycle
The fast cycle, as the name suggests, encompasses those aspects of the carbon exchange which happen more quickly. This includes carbon that is passed between living organisms, and carbon exchanges between living things and the atmosphere. In this way, the fast cycle is measured in lifespan: the lifespan of any given organism, be that plant or animal.
Photosynthesis
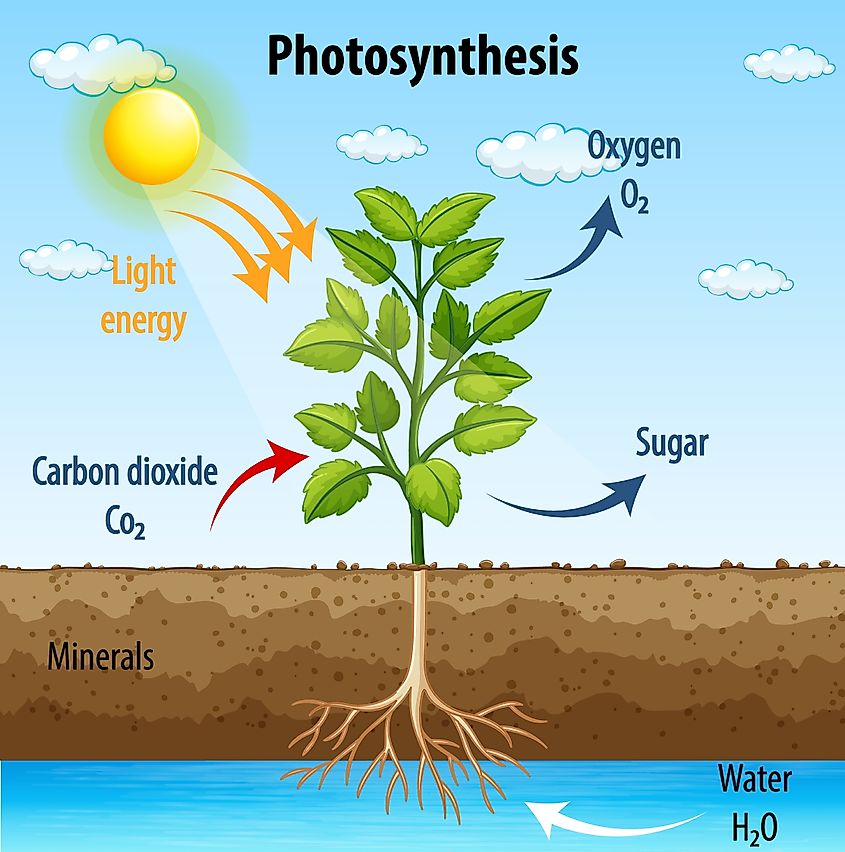
Carbon which exists in the atmosphere, in carbon dioxide (CO2) form, is absorbed by plants. Plantlife uses carbon dioxide, along with sunlight and water, and transforms them into sugars, in a process called photosynthesis. The carbon is pulled from the air via stoma on a plant's leaves. This then interacts with sunlight and water to provide the energy a plant needs to grow. Oxygen and sugar are products of this process. This is why plants are so important for human life as they provide oxygen. Without carbon, this process could not exist.
Consumption

A plant can also transfer its carbon to animals when it is consumed. When a plant is eaten, its energy reserves are then transferred to that creature, and so too are the carbon atoms within the plant. This is the second part of the fast cycle. Carbon moves its way through a food chain in any given ecosystem, as smaller prey animals are eaten by predators higher in the food chain. The carbon remains within the food chain until it is released through decomposition following the death of a plant or animal, or through respiration.
Respiration

As mentioned above, plants take in CO2 and release oxygen (O2). Animals, including humans, breathe in this O2. The process of respiration in humans and animals involves these O2 molecules bonding with carbon, to produce CO2. The respiration process, which we usually think of simply as breathing, provides the necessary oxygen to our bloodstream and bodies and creates a CO2 byproduct, which we then breathe out, or release back into the atmosphere. In this way, carbon moves easily from the atmosphere to a plant, to an animal, and then back to the air. The carbon is constantly in motion within this fast carbon cycle. Roughly 1,000 to 100,000 million metric tons of carbon move through the fast carbon cycle every year.
Decomposition

Decomposition occurs after a plant or animal dies. Once it is deceased, bacteria break down the material from these organisms, breaking the bonds between these strong carbon compounds. As these bonds are broken, CO2 is released back into the atmosphere. In this way, the carbon completes a full cycle, moving from the air to plants and/or animals, and is returned to the atmosphere.
Slow Cycle
While a large amount of carbon circulates through the fast cycle, there is another aspect of the carbon cycle known as the Slow Cycle. In this cycle, carbon is exchanged at a much slower rate, over thousands and millions of years, between the atmosphere and the lithosphere, which primarily includes the Earth’s crust. Much of the rocks and minerals in the Earth’s crust contain carbon, or carbon compounds.
Acid Rain
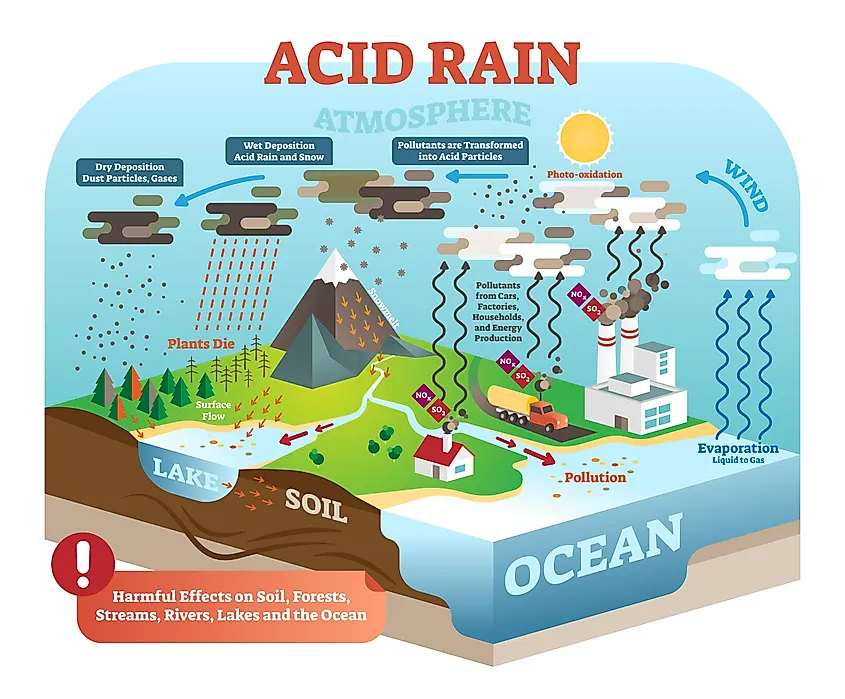
The exchange of carbon from the atmosphere to the earth happens in a few different ways. One of these ways is through rainwater. CO2 accumulates in the air and saturates rainwater to form a mild type of acid rain. The acidic nature of this rainwater causes it to eat away at solid rock, dissolving and eroding it over time. As the rocks are weathered, minerals are released, which in turn create inorganic carbons. Carbon and acid rainwater can also land in the ocean. When water with a high carbon content hits the ocean, it dissolves and produces bicarbonate ions. This can then bond with calcium to create the compound known as calcium bicarbonate, an essential component in the makeup of shells and many crustaceans. Shells that sink to the bottom of the seafloor (for example, when shellfish die) will eventually, with time and pressure, compact and merge into the ocean bed, becoming part of the lithosphere.
Decomposition And Fossil Fuels

In a similar way, organic life can become trapped in mud, sand or bogs, when they die and begin to decompose. In normal decomposition, carbon bonds are broken down by bacteria and released back to the atmosphere. But in other cases, the organism does not decompose in an aerobic fashion, and the carbon becomes trapped. Great pressure over time will compress and trap these decomposing carbon-based life forms, creating pockets of carbon-based fuel, known as fossil fuels. These fuels can be released from the lithosphere naturally when tectonic plates shift, or, more commonly, when they are mined and burned as fuel by humans. In this way, the carbon goes through a much slower cycle, but still circles from the atmosphere, into the ground, and eventually back again.











